Latest News

Three Amherst Regional Middle School counselors absolved of Title IX offenses
AMHERST — Even with complaints that their actions and behavior, including intentional misgendering students were likely offensive, three counselors at the Amherst Regional Middle School have been cleared of violations of the federal Title IX law in...

Photos: Magnolia maintenance
Most Read
 ‘Our hearts were shattered’: Moved by their work in Mexico soup kitchen, Northampton couple takes action
‘Our hearts were shattered’: Moved by their work in Mexico soup kitchen, Northampton couple takes action
 Springfield man charged with murder in Holyoke stabbing
Springfield man charged with murder in Holyoke stabbing
 Amherst-Pelham schools look to address school absences with new plan
Amherst-Pelham schools look to address school absences with new plan
 Two men dump milk, orange juice over themselves at Amherst convenience store
Two men dump milk, orange juice over themselves at Amherst convenience store
 Next 5-story building cleared to rise in downtown Amherst
Next 5-story building cleared to rise in downtown Amherst
 Three Amherst Regional Middle School counselors absolved of Title IX offenses
Three Amherst Regional Middle School counselors absolved of Title IX offenses
Editors Picks
 All about conviction: PVPA team wins state, heads to nationals of Mock Trial Championship
All about conviction: PVPA team wins state, heads to nationals of Mock Trial Championship
 Spring brings new art: A look at what's on tap in April at selected local galleries
Spring brings new art: A look at what's on tap in April at selected local galleries
 News Briefs: Roadwork in Plainfield; bake sale in Hadley
News Briefs: Roadwork in Plainfield; bake sale in Hadley
 Springfield man charged with murder in Holyoke stabbing
Springfield man charged with murder in Holyoke stabbing
Sports

High school: Northampton softball slugs past Mahar for season's first victory (PHOTOS)
Following four frustrating losses to start the season, including three straight in five innings, the Northampton softball team was on the other side of a mercy-rule contest on Monday afternoon.The Blue Devils scored at least three runs in each of the...
 Baseball: Smith Academy can't overcome early deficit in 13-6 loss to Mahar
Baseball: Smith Academy can't overcome early deficit in 13-6 loss to Mahar
Opinion

Guest columnist Jay Fleitman: Can’t leave Hamas intact
The editorial space of this newspaper is replete with demands for a cease-fire in Gaza, and similar demands have been made by the governing bodies of Northampton and Amherst. This really is a “demand” for the Israelis to unilaterally stop their...
 Walter Krzeminski: Locals who homered at Fenway for the home team
Walter Krzeminski: Locals who homered at Fenway for the home team
 Mary Collins: Let’s create 413 Day
Mary Collins: Let’s create 413 Day
 Judy Gutlerner: More smiles
Judy Gutlerner: More smiles

Business

Tea Guys of Whately owes $2M for breach of contract, judge rules
WHATELY — Tea Guys LLC must pay more than $2 million to a Baltimore tea company due to a breach of contract, a Franklin County Superior Court judge has ruled.The small business in Whately was sued last summer by Zest Tea LLC and had two bank accounts...
 Black business group presses demand for Amherst relief funds
Black business group presses demand for Amherst relief funds
Arts & Life

Speaking of Nature: Indulging in eye candy: Finally, after such a long wait, it’s beginning to look like spring is here
I have just about reached the end of my patience with the winter of 2024. I realize that this may sound a bit strange, especially because we are now in the beginning of spring, but those of us who bore the brunt of the April snowstorm may sympathize...
Obituaries
 Virginia Seney
Virginia Seney
LEEDS, MA - Friendly Ice Cream Corporation announces the slashing of their coffee ice cream production after the passing of Virginia Ann Heon Seney on April 13, 2024 at the age of 89. Born ... remainder of obit for Virginia Seney
 Elizabeth A. O'Brien-Means
Elizabeth A. O'Brien-Means
Williamsburg, MA - Elizabeth Ann O'Brien-Means, 71, of Briar Hill Rd. passed peacefully on Thursday at the Senior Living Campus at Linda Manor in Leeds. Betsy, as family and friends called ... remainder of obit for Elizabeth A. O'Brien-Means
 Kathleen A. Dunn
Kathleen A. Dunn
Northampton, MA - Kathleen Ann (Spellman) Dunn, 79, of Northampton, MA, passed away on April 8, 2024 surrounded by family, at Community Hospice House in Merrimack, NH. She was born in P... remainder of obit for Kathleen A. Dunn
 Joseph Walter Walas Jr.
Joseph Walter Walas Jr.
Joseph Walter Walas, Jr. East Haddam, CT - Joseph Walter Walas, Jr., 73, of East Haddam, formerly of Durham, and husband of the late Linda (Good) Walas, passed away on Wednesday, March 27, 2... remainder of obit for Joseph Walter Walas Jr.

 Wheeling for Healing returns to South Deerfield to raise money for cancer treatment
Wheeling for Healing returns to South Deerfield to raise money for cancer treatment
 Sadiq to leave Amherst middle school principal role
Sadiq to leave Amherst middle school principal role
 Northampton seeks bids to redevelop former Registry of Deeds property
Northampton seeks bids to redevelop former Registry of Deeds property
 Hadley’s Brad Mish, Northampton’s Elianna Shwayder top Hampshire County finishers at 128th Boston Marathon
Hadley’s Brad Mish, Northampton’s Elianna Shwayder top Hampshire County finishers at 128th Boston Marathon
 Softball: South Hadley’s Ella Schaeffer records 500th career strikeout in loss to East Longmeadow
Softball: South Hadley’s Ella Schaeffer records 500th career strikeout in loss to East Longmeadow
 Columnist Razvan Sibii: How to welcome a refugee family into your community
Columnist Razvan Sibii: How to welcome a refugee family into your community
 Lawmakers, Jewish groups accuse Massachusetts Teachers Association of bias
Lawmakers, Jewish groups accuse Massachusetts Teachers Association of bias
 Homeless man gets three years for Northampton rape
Homeless man gets three years for Northampton rape
 High schools: Holyoke boys volleyball sweeps Athol for 5th straight victory (PHOTOS)
High schools: Holyoke boys volleyball sweeps Athol for 5th straight victory (PHOTOS) More history for Tiger Woods. He makes the Masters cut for a record 24th time in a row
More history for Tiger Woods. He makes the Masters cut for a record 24th time in a row Track & field preview 2024: Not happy with last year’s performance, Northampton boys seeking redemption
Track & field preview 2024: Not happy with last year’s performance, Northampton boys seeking redemption Columnist Sara Weinberger: The appalling silence over the atrocities of Oct. 7
Columnist Sara Weinberger: The appalling silence over the atrocities of Oct. 7 Recognizing an ‘inspiring force’: City business owner honored with Black Excellence award
Recognizing an ‘inspiring force’: City business owner honored with Black Excellence award Consumer Corner with Anita Wilson: A two-day reprieve in tax filing deadline offers time for tips
Consumer Corner with Anita Wilson: A two-day reprieve in tax filing deadline offers time for tips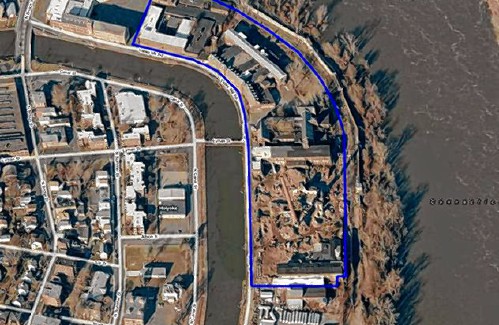 Sublime Systems lands $87M federal award for low-carbon cement plant in Holyoke
Sublime Systems lands $87M federal award for low-carbon cement plant in Holyoke Weekly Food Photo Contest: This week’s winner: Nicholas Horton of Northampton
Weekly Food Photo Contest: This week’s winner: Nicholas Horton of Northampton What does freedom look like today? On view at Williams College, seven Black American artists interpret the meaning of emancipation
What does freedom look like today? On view at Williams College, seven Black American artists interpret the meaning of emancipation Book Bag: ‘Dear Oliver: An Unexpected Friendship With Oliver Sacks’ by Susan B. Barry; ‘Benjy’s Messy Room’ by Barbara Diamond Goldin
Book Bag: ‘Dear Oliver: An Unexpected Friendship With Oliver Sacks’ by Susan B. Barry; ‘Benjy’s Messy Room’ by Barbara Diamond Goldin Only Human with Joan Axelrod-Contrada: To journal or not to journal: Advice for when journaling feels like it’s holding you back
Only Human with Joan Axelrod-Contrada: To journal or not to journal: Advice for when journaling feels like it’s holding you back
